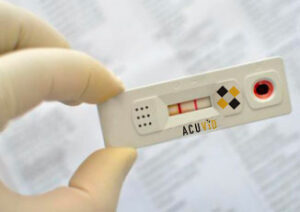
Toronto, Ontario–(Newsfile Corp. – March 25, 2021) – Therma Bright Inc. (TSXV: THRM) (“Therma” or the “Company”), developer of the AcuVid™ Covid-19 Rapid Antigen Saliva Test and other progressive diagnostic and medical device technologies, is pleased to announce that it has received IRB approval and subsequently submitted to Health Canada seeking approval to conduct a clinical performance study of its AcuVidTM Covid-19 Rapid Saliva-based Antigen Test.
Therma’s clinical research organization prepared and filed the necessary documentation for the IRB and Health Canada submissions. They are now focused on readying the trial site and personnel to begin the study once the Health Canada go ahead is received.
Rob Fia, CEO, commented, “This is yet another significant milestone in moving our AcuVid™ test closer to market. We look forward to beginning the trial in Ontario, Canada as we explore other potential sites in the U.S., South America and Europe that will generate additional data to support our applications in those jurisdictions.”
Last week, the US FDA announced new guidance to streamline the approval and use of rapid Covid-19 tests in the US.
Therma Bright believes its AcuVidTM COVID-19 Rapid Antigen Saliva Test ideally meets the new March 16 FDA Guidance for test developers seeking emergency use authorization (EUA) of certain tests for screening using serial testing. Serial testing involves testing the same individual multiple times within a few days and can increase chances of detecting asymptomatic infection that might not otherwise show up with a single test. CDC recommends serial testing at least once per week, along with other mitigation measures, such as masking and social distancing, to reduce disease transmission. In addition, with the recent American Rescue Bill passed by President Biden, there are opportunities to assist the U.S. and its citizens detect, contain and mitigate COVID-19 and its growing variants with Therma Bright’s “Made in the USA” AcuVidTM COVID-19 Rapid Antigen Saliva Test once approved under the Emergency Use Authorization (EUA) by the FDA.
Mr. Fia commented, “This FDA action should positively impact the approval of our test in the U.S. and accelerate the acceptance and use of AcuVidTM which is ideally suited for serial testing due to its ease of use and rapid results.”
Therma Bright is not making any express or implied claims that its test product has the ability to eliminate or cure COVID-19 or the SARS-CoV-2 virus.